Pharmaceutical
We keep pharmaceutical manufacturers, biologics manufacturers, and compounding facilities safe from cross contamination and harmful microorganisms.
About The Invisible Enemy
Bacteria and viruses are stronger than ever. They travel faster, farther, and are becoming increasingly resistant to antibiotics and traditional disinfection techniques. Much of what once worked to combat these dangerous microbes becomes less effective with every passing day.
The pharmaceutical industry faces increasing pressure to ensure the integrity of their products and maintain facilities that are free from contamination of any kind. Failure to maintain good manufacturing practices (CGMC) has proven to have devastating consequences, from life threatening health risks, to a companies’ reputation, to the bottom line. Pharmaceutical manufacturers must find a way to keep their facilities sterile and protected from dangerous microorganisms.
Ecosense Offers A Practical Solution
Enter Ecosense. We provide pharmaceutical manufacturers using aseptic processing or terminal sterilization methods access to the world’s most modern decontamination, sterilization and infection prevention technologies. Our treatments target and kill the spores, bacteria, viruses and other microorganisms that pose a threat to your manufacturing process. We build a custom tailored decontamination plan that protects your business, and do so in a way that is verifiable and affordable.
Ecosense decontamination and sterilization programs include the following:
Contact us today to see how we can help keep your commercial or government building safe from harmful microorganisms. We offer free testing and risk assessments.
Effective
We can achieve a 99.9999% bio-burden reduction of spores, bacteria, viruses and fungi hiding in difficult to reach areas.
Fast
Our treatments are applied quickly and efficiently, guaranteeing minimal downtime at your facility.
Safe
Our preferred products have low toxicity ratings without harmful chemical residuals, which helps keep everyone safe.
Affordable
Our services are affordable and significantly less expensive than the cost of a recall or outbreak.
Pharmaceutical Manufacturing Facility Decontamination
Pharmaceuticals
At Ecosense, we work with pharmaceutical companies and biosimilar manufacturers that operate under USP and ISO guidelines. Contract manufacturers and companies that engage in multi-product manufacturing use our services to avoid the risk of cross contamination that increases exponentially with each product change.
With Ecosense, you select from proven pharmaceutical decontamination technologies, from ClorDisys’ gaseous chlorine dioxide system to MarCore’s dry fogging system, which have earned worldwide recognition in the pharmaceutical industry. From sterilizing facilities as large as 4,400,000 Cubic Feet to re-purposing single pieces of equipment contaminated with beta-lactams, we’ve got you covered.
For pharmaceutical companies, the consequences of an adverse biological occurrence at a manufacturing plant can be devastating and include:
Contact us today to learn how we can help prevent the spread of harmful contamination at your pharmaceutical company.


Biologics Manufacturers
Biologic drug manufacturers face unique challenges compared to traditional pharmaceutical manufacturers, and a chief concern is to maintain a facility free from contamination of any kind. Because biologic drugs are manufactured inside living systems, including plant and animal cells, it is essential that these living systems remain sterile and avoid contamination.
The margin of error is slim and the risk of contamination is a risk that most companies cannot afford to take. At Ecosense, we help minimize your risk and keep your biologics facility free from contamination through a tailored sterilization plan that’s right for your business.
Contact us today to get a free swab test and risk analysis.
Compounding Facilities
Pharmaceutical compounding is the creation of a particular pharmaceutical product to fit the unique need of a patient. Because each product is manufactured to fit the specific requirements of an individual, it is imperative that the process remains free from contamination from harmful microorganisms of any kind. Failure to do so could result in a compromised product, the consequences of which could be devastating. The sheer volume of constantly changing orders significantly complicates the responsibility of maintaining a contamination free environment. We have a better idea.
Contact us today to claim your free swab test and risk anslysis.

Our Decontamination and Prevention Services Target:
Bacterial Endospores
Myco Bacteria
Small Non-Enveloped Viruses
Fungi & Mold
Gram (-) Negative Bacteria
Gram (+) Positive Bacteria
Large Non-Enveloped Viruses
Enveloped Viruses
Beta Lactums & Pinworms
From the easiest to destroy lipid viruses to the most difficult to kill bacterial spores, we craft a custom tailored plan which will protect your business from harmful contamination and save you time & money
Tailored Decontamination and Protection Plans
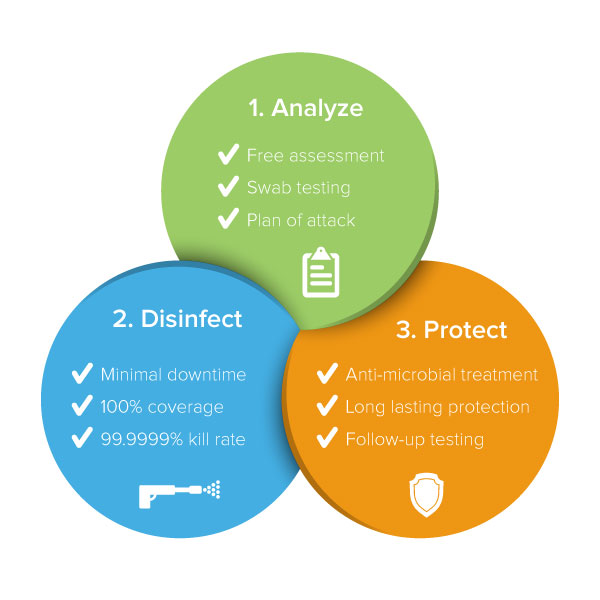
Free Assessments
We offer free assessments of your exposure to harmful contamination by measuring the contamination levels at various locations throughout your facility. Based on your risk assessment, we put together a plan to help your facility fight harmful contamination and the spread of infection.
Decontamination
We decontaminate your facility using state of the art technologies and target dangerous microorganisms wherever they reside.
Long-Term Surface Protection
Our anti-microbial treatments, when applied after disinfection, provide an ongoing layer of protection against the invisible enemy. We’ll take follow up readings every 3 months to help ensure you’re protected over time. We’ll also train and work with your staff to ensure they’re doing all they can to reduce the threat of contamination.
Contact us today to see how we can help keep your pharmaceutical facility safe from infectious disease outbreaks.




